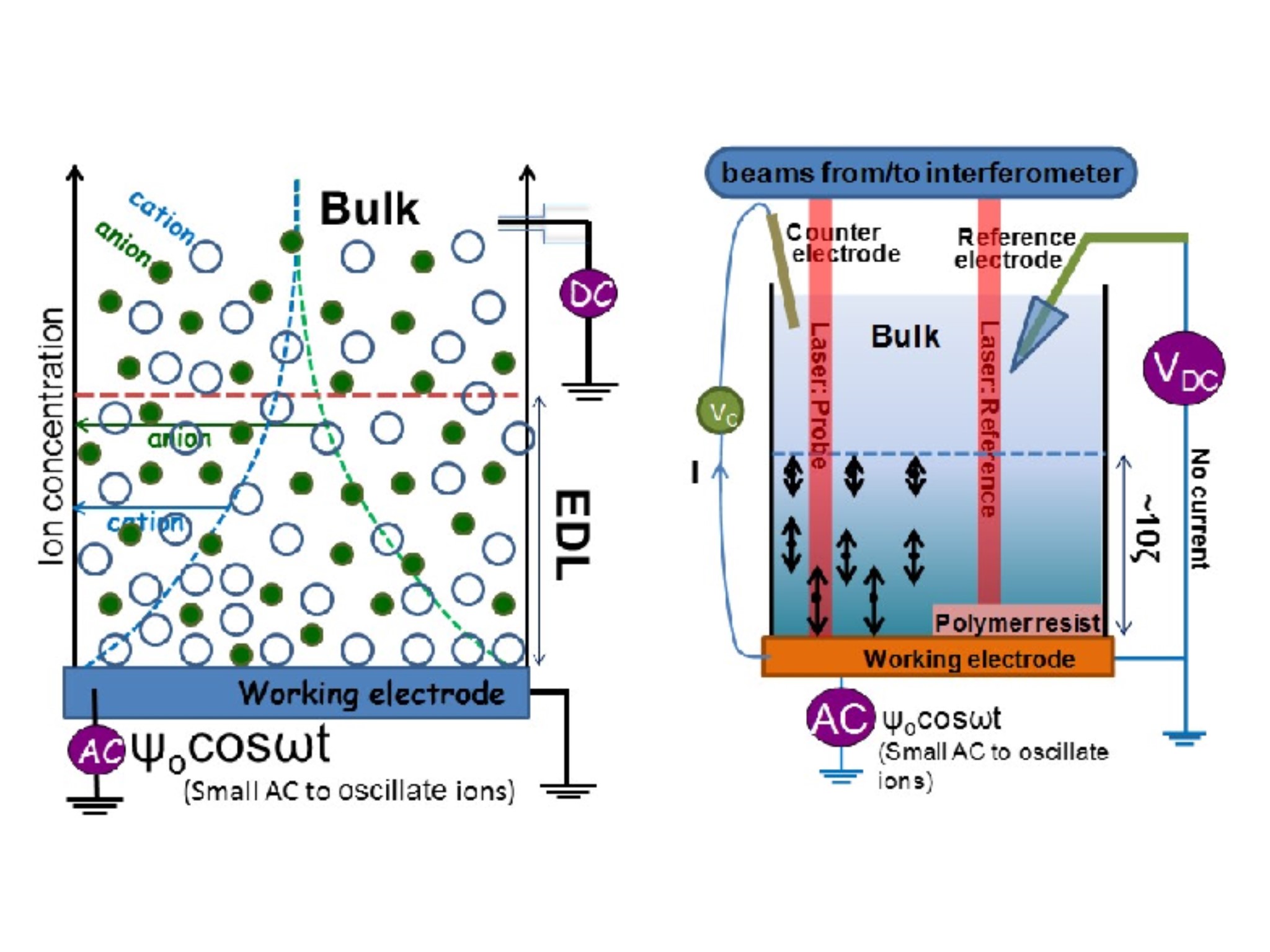
Electrical Double Layer based devices
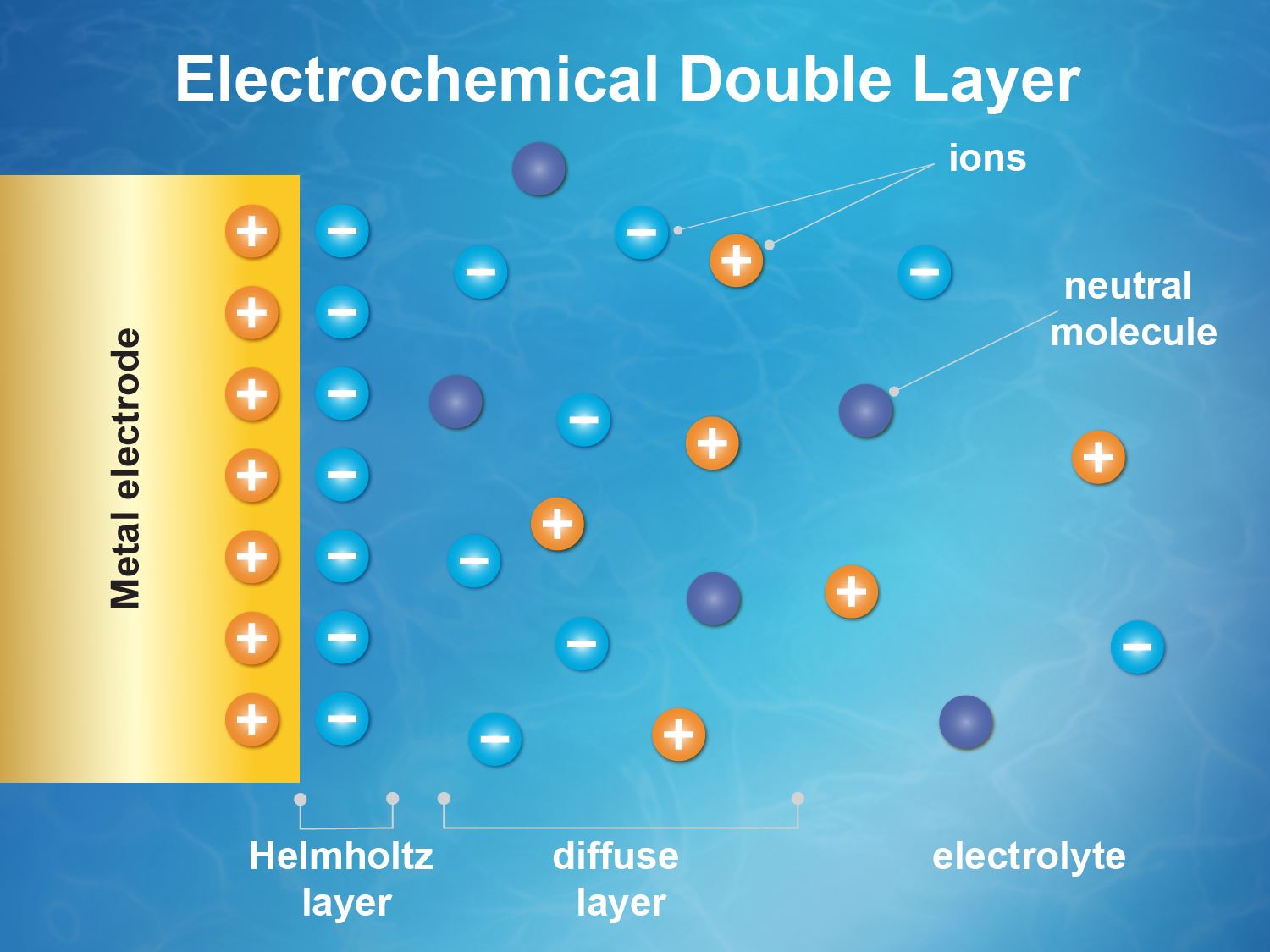
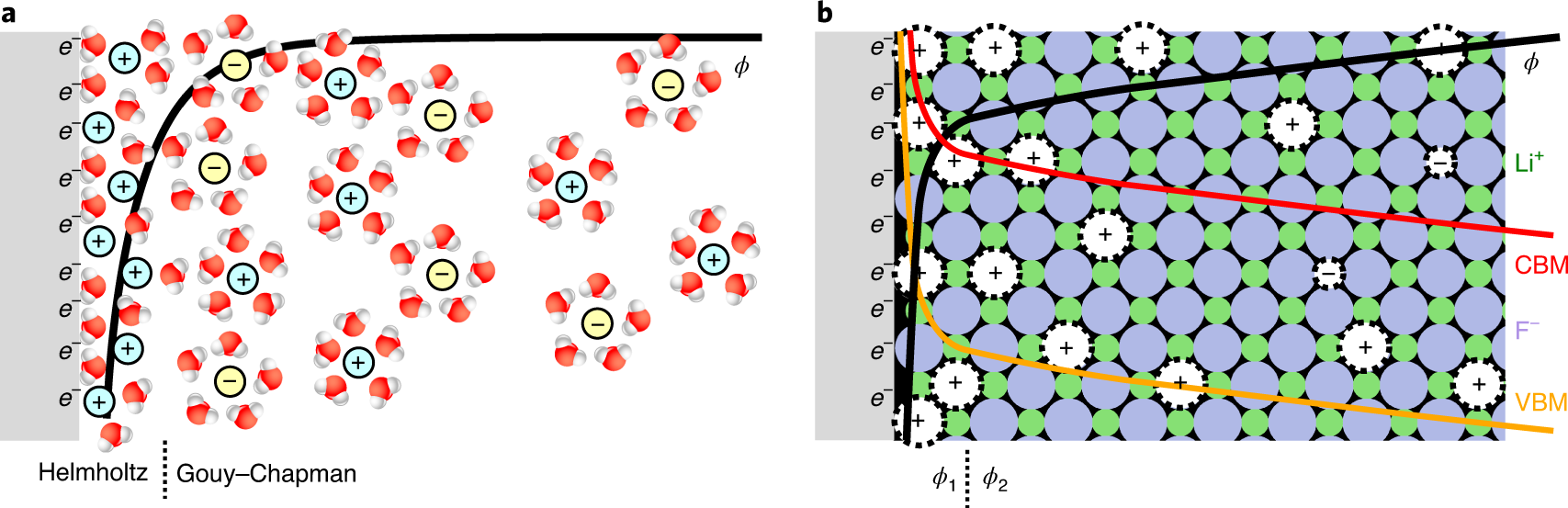
Immersing a metal (electrode) in a salt solution will spontaneously accumulate ions due to image charge to form the well known electrical double layer (EDL). Sign of the charge depends on the Fermi level (i.e., the electrochemical potential) difference between the metal and the solution. For example, gold will accumulate negative ions (i.e., anions). The interfacial structure

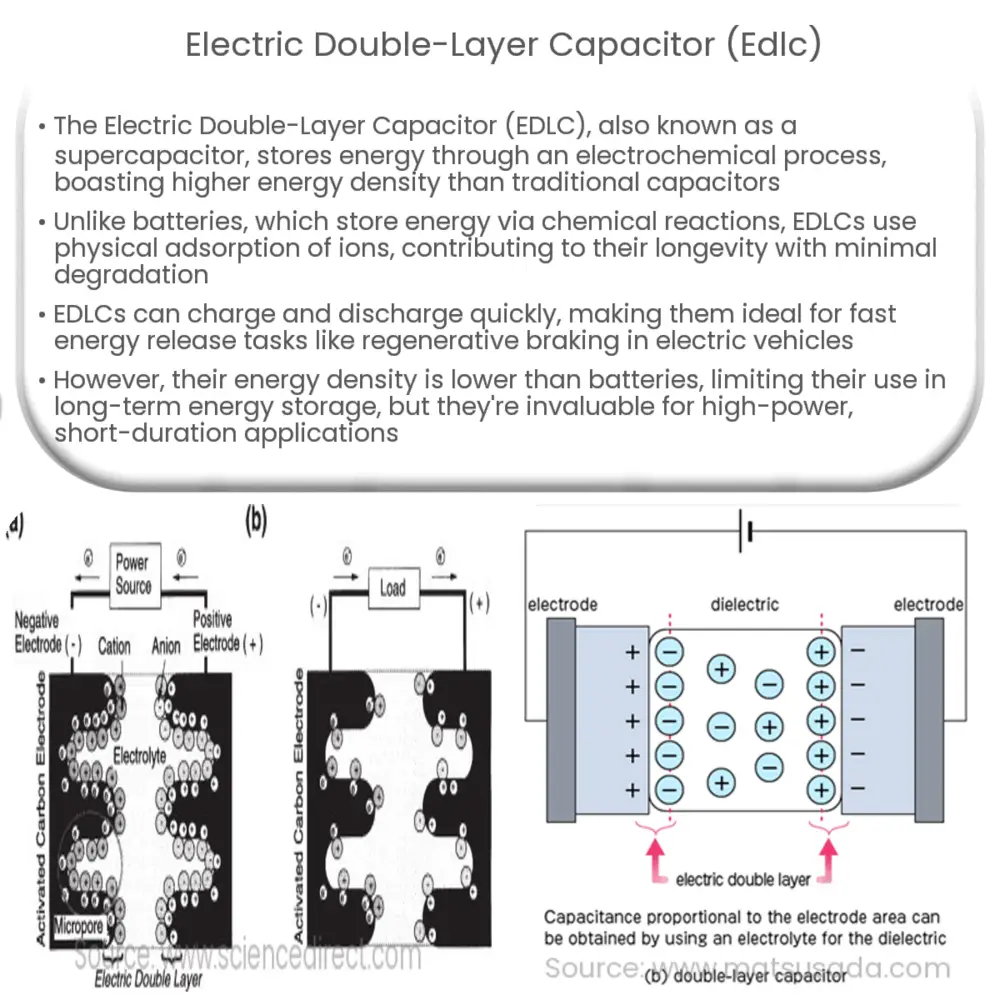
Combination of two Double Layer Capacitor electrode with a solvent based ionic layer.
Insights into the electric double-layer capacitance of two-dimensional electrically conductive metal–organic frameworks - Journal of Materials Chemistry A (RSC Publishing)

Electrical Double Layer Based Devices College Of, 41% OFF

ChemRxiv on X: Modeling the Electrical Double Layer to Understand the Reaction Environment in a CO2 Electrocatalytic System by Wilson Smith & co-workers / X
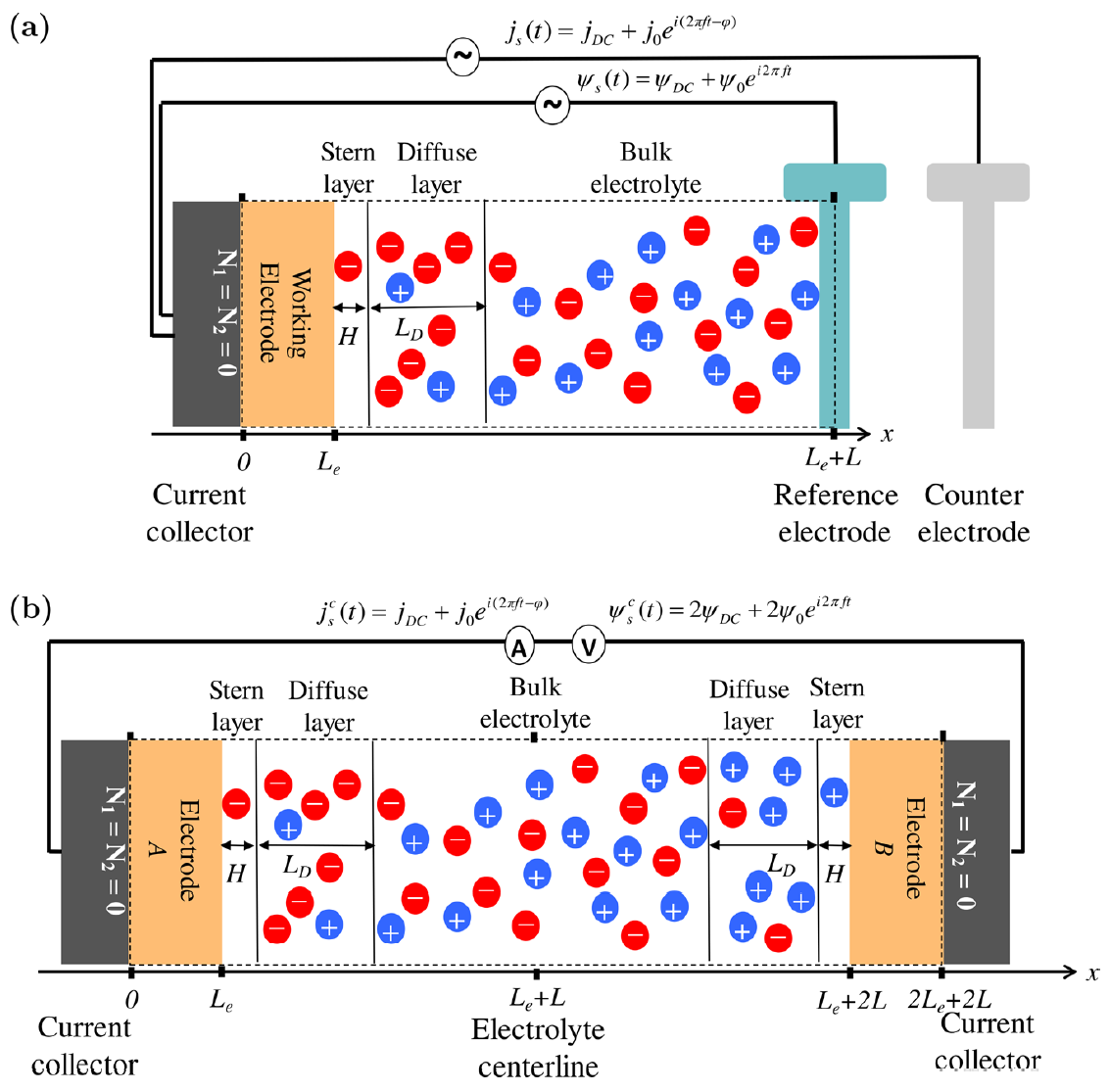
Physical Interpretations of Nyquist Plots for EDLC Electrodes and Devices
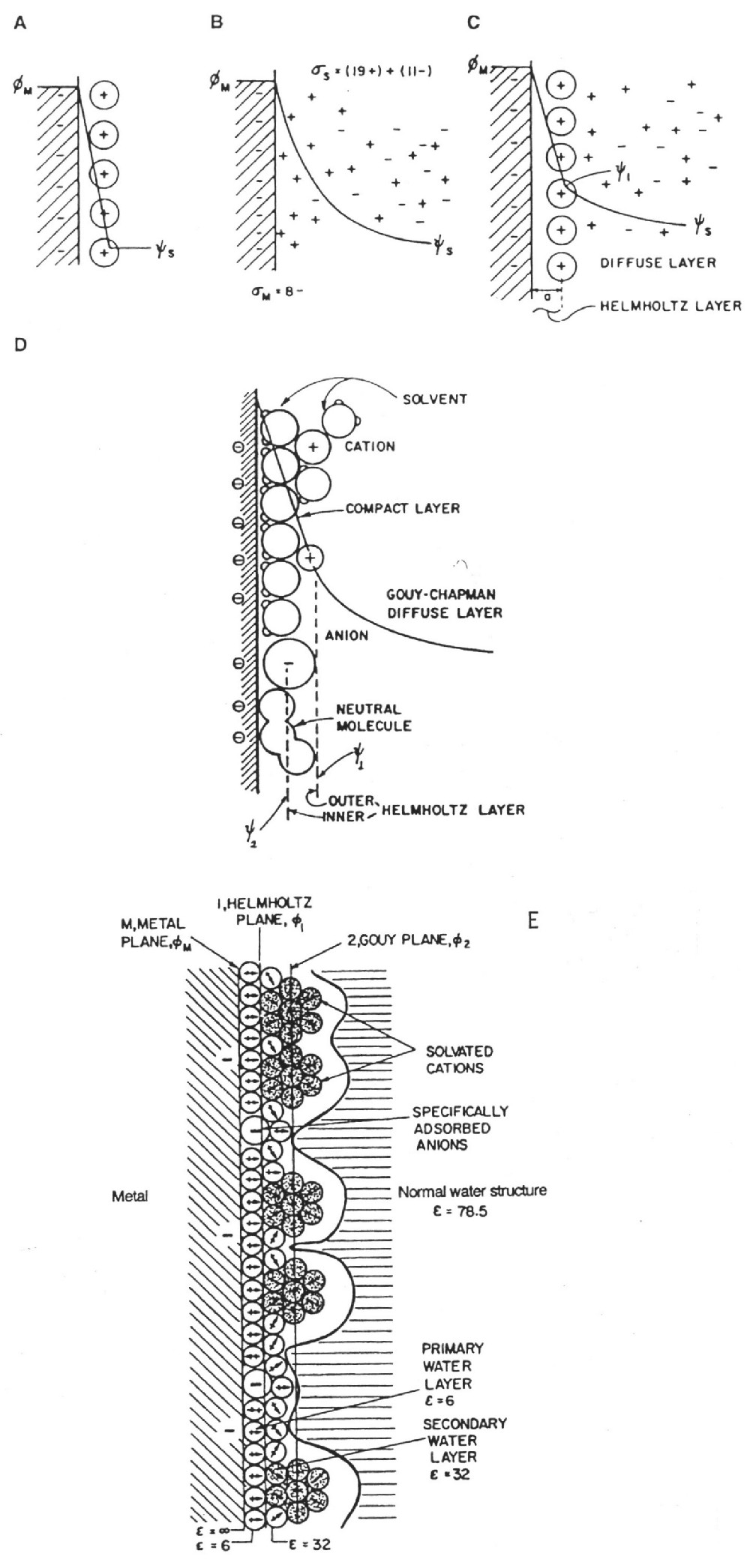
Electrochemistry Encyclopedia -- Electrochemical capacitors

Electrical Double Layer Based Devices College Of, 41% OFF
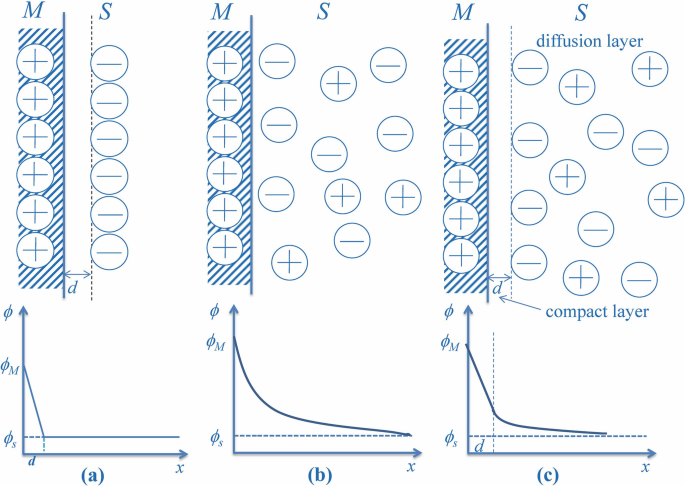
Electrical Double-Layer Structure and Property of Ionic Liquid-Electrode System for Electrochemical Applications
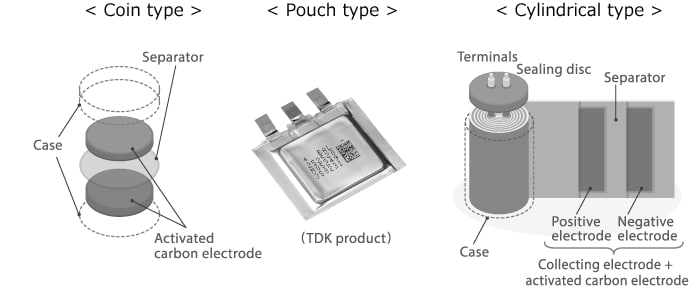
Principles of EDLCs|TDK Techno Magazine|Electronics ABC|Learn about Technology with TDK