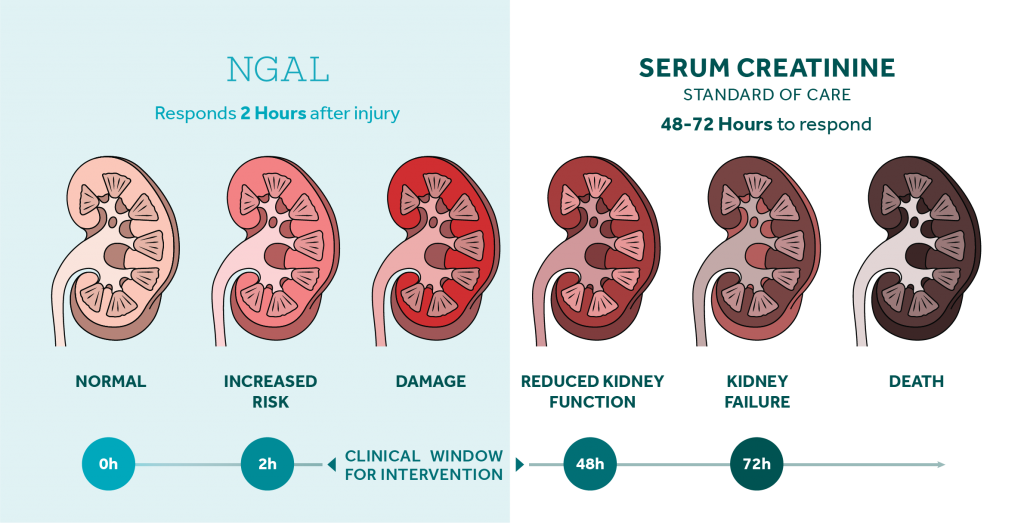
NGAL - Bioporto
The NGAL Test is a particle-enhanced turbidimetric immunoassay for the quantitative determination of NGAL in human urine and plasma on automated clinical chemistry analyzers. NGAL measurements are useful in the risk assesment of AKI.

About BioPorto - Bioporto

PDF) Cost-effectiveness and value of information analysis of NephroCheck and NGAL tests compared to standard care for the diagnosis of acute kidney injury

Plasma orexin A does not reflect severity of illness in the intensive care units patients with systemic inflammation, JA Clinical Reports

NGAL - Bioporto

Product Details - Bioporto

BioPorto Diagnostics A/S: Contact Details and Business Profile

/wp-content/uploads/2023/12/home-hero

Elevated Neutrophil Gelatinase-Associated Lipocalin Is Associated With the Severity of Kidney Injury and Poor Prognosis of Patients With COVID-19 - ScienceDirect

BioPorto Diagnostics A/S

Ulcer-associated cell lineage expresses genes involved in regeneration and is hallmarked by high neutrophil gelatinase-associated lipocalin (NGAL) levels. - Abstract - Europe PMC

Full article: Establishment of Acute Kidney Injury Mouse Model by 0.75% Adenine Ingestion

The NGAL Test Reagent Kit - Bioporto








