Preparation of Standard Solution of Sodium Carbonate - Chemistry
A common primary standard for standardizing strong acids is sodium carbonate (Na2CO3).For acid-base titration, it is customary to prepare solutions of an acid and base of the desired concentration. Visit BYJU
A common primary standard for standardizing strong acids is sodium carbonate (Na2CO3).For acid-base titration, it is customary to prepare solutions of an acid and base of the desired concentration. Visit BYJU'S to understand more about it.

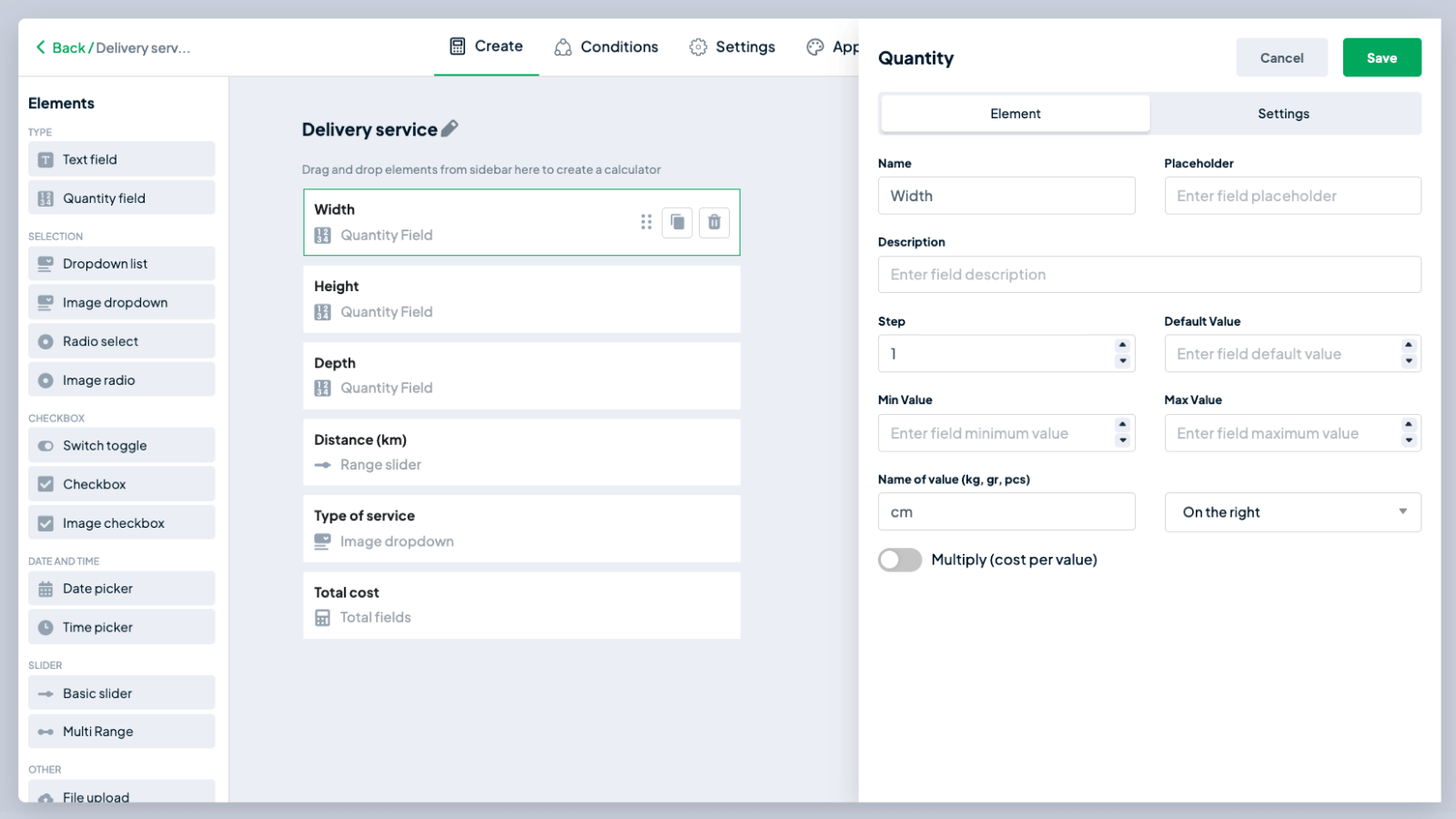
Preparing a standard solution - sodium carbonate

SOLUTION: 13 must know experiments in chemistry - Studypool

Page 9 - HKDSE CHEMISTRY - A Modern View (Second Edition)

GyanPub on Tumblr: Prepare M/10 sodium carbonate solution Prepare
Acids-Bases and Salts-Volumetric analysis, Chemistry tutorial

Preparation of Standard Solution of Sodium Carbonate: Theory

How do you prepare a standard solution? - A Plus Topper

Title: Lesson 14 Preparing a Standard Solution and Back Titration

Preparing a standard solution - sodium carbonate

experiment four practicals - Preparation of a standard solution of
qph.cf2.quoracdn.net/main-qimg-344f97f56ef5d8d6b96

Preparing a standard solution - sodium carbonate

PDF) Chemistry Experiment Laboratory Report (1) Title

Conversion of gaseous effluents of power plant to sodium carbonate