The value of compression factor at the critical state of a vander waals gas is
The value of compression factor at the critical state of a vander waals gas is

Compressibility Factor of Gas, Overview, Equation & Chart - Lesson

Telugu] The compression factor (compressibility factor) for one mole

Compressibility factor (Z) for a van der Waals real gas at critical po

108. Which of following statement (s) is true 1 - Slope of isotherm critical point is maximum. 103. 11 - Larger is the value of T, easier is the liquification of gas.X
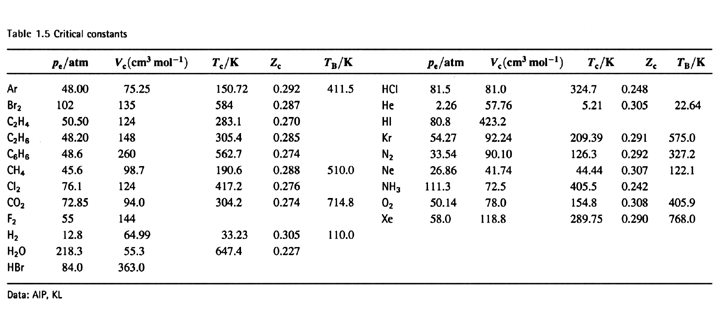
Chapter 1 Properties of Gases
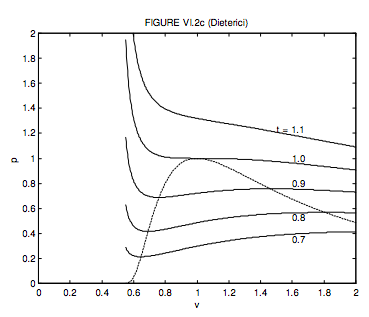
6.3: Van der Waals and Other Gases - Physics LibreTexts

SOLVED: Text: critical point; the critical parameters can Using the isotherm curve of van der Waals equation at the be expressed in terms of van der Waals coefficients and b: Tc =

Rarefaction shock region (RSR) for a van der Waals polytropic gas

Van der Waals Equation:Calculate the expansion and compresibility
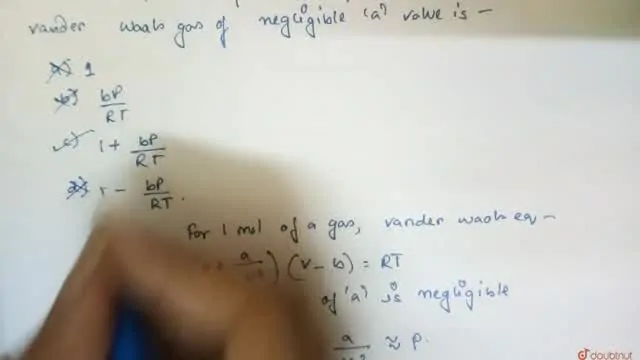
Bengali] The compresibility factor (Z) of one mole of a van der waals

The properties of gases - Wissenschaft Online

8.6: Non-Ideal Gas Behavior General College Chemistry I

SOLVED: The following equation of state is known as the Dieterici equation: p(Vm - b) = RT * e^(RT/Vm), where a and b are very small constant values. Show that at low

Bengali] What will the value of compressibility factor (Z) be for a g

PDF) Critical State Behavior of Van der Waal gases Conformation to Nelson Obert Characteristics














